Lung Ultrasound And COVID-19
In the fight against COVID-19, healthcare providers face challenges identifying and evaluating the management of patients who have contracted the coronavirus. Chest x-rays and CT scans are traditional methods for identifying pulmonary changes that may occur in patients with COVID-19, but there are other quick and effective options, such as lung ultrasound.
To help teach medical professionals how to utilize lung ultrasound, we’ve provided FREE access to our Lung Ultrasound Lesson below. Though typically only accessible with a paid subscription to Block Buddy Pro, we understand the importance of ultrasound technology in the fight against COVID-19. As a result, we made the Lesson widely available to help assist healthcare providers in detecting pulmonary changes in conjunction with lab results and clinical findings.
Why use Lung Ultrasound?
The use of lung ultrasonography is being employed by healthcare providers across the globe to help screen and evaluate the management of COVID-19 patients. Due to its small size and portability, an ultrasound machine can be quickly utilized at the bedside. Within minutes, a lung ultrasound scan can be completed. This prevents the transport of a suspected or confirmed COVID-19 patient to the CT scanner, and avoids potential exposure of other patients and staff members to the virus. It also avoids the process of disinfecting the CT scanner and ensuring air within the CT room has been properly exchanged. Compared to a traditional chest x-ray, lung ultrasound avoids radiation exposure and is more sensitive in detecting various lung pathologies. Given the current climate with COVID-19, lung ultrasound is a safe, efficient, and effective tool used in the treatment of COVID-19 patients.
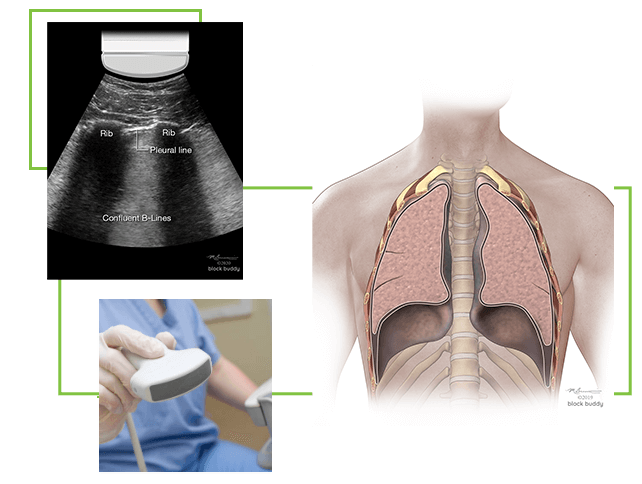
COVID-19 and Lung Ultrasound
- COVID-19 ultrasound findings (B-lines, consolidations, air bronchograms, and pleural abnormalities) are most commonly seen in the posterior lower fields of both lungs.
- Ultrasound is a quick and efficient tool that can be used at the bedside to detect pulmonary changes that may occur in COVID-19 patients.
- CXR and chest CT are non-specific and expose the patient to radiation.
- Ultrasound scans can be repeated easily to assess disease process.
- Due to the small size of the COVID-19 virus, the particles may be inhaled into the lungs and alveoli.
- Infection usually starts in the alveoli, close to the pleura, allowing for easy identification on ultrasound.
- Perform the ultrasound scan using a low frequency curvilinear or phased array transducer.
- In addition, a high-frequency linear transducer can be used to improve image resolution and enhance diagnostic accuracy of pleural abnormalities and parapneumonic effusions.
- The findings below are based on current knowledge and understanding of COVID-19.
- The findings may be variable, depending on the severity/stage of the disease process.
Mild COVID-19
- Normal or reduced pleural sliding
- Regular or mild pleural thickening
- Focal B-lines
- Mild pulmonary edema
Moderate to Severe COVID-19
- Pleural line is thickened, irregular and interrupted
- Multiple or confluent B-lines
- Subpleural consolidation may appear patchy or nodule
- Air bronchiologram or air bronchogram may be present within consolidation
- Color Doppler may show insufficient blood supply within consolidation
- Moderate to severe alveolar-interstitial pulmonary edema
- Large pleural effusion (rare)
Useful Links:
- Findings of Lung ultrasonography of novel corona virus pneumonia during the 2019-2020 epidemic.
- A preliminary study on the ultrasonic manifestations of peripulmonary lesions of non-critical novel coronavirus pneumonia.
- Can Lung US Help Critical Care Clinicians in Diagnosis of Novel Coronavirus (COVID-19) Pneumonia?
- Point-of-Care Lung Ultrasound findings in novel coronavirus disease-19 pnemoniae: a case report and potential applications during COVID-19 outbreak
- Proposal for international standardization of the use of lung ultrasound for COVID-19 patients; a simple, quantitative, reproducible method.
- COVID-19 outbreak: less stethoscope, more ultrasound.
Lung Ultrasound Lesson
Due to the current pandemic and the importance of lung sonography in providing care to patients across the globe, we are happy to provide free access to the newly expanded lung ultrasound lesson from the Block Buddy Pro app. Find other lessons on Point-of-Care Ultrasound techniques, Peripheral Nerve Blocks, and more on Block Buddy Pro, available now in the App Store and Google Play Store.
In This Lesson
- Equipment
- Indications
- Anatomy
- Positioning and Scanning
- Normal Lung Ultrasound
- Abnormal Lung Ultrasound
- Pulmonary Pathology
- Clinical Pearls
- Video Gallery
- Illustrations
- References
1. Equipment
- High frequency linear transducer (6-13MHz), low frequency curved transducer (2-5 MHz), or phased array transducer (1-5 MHz).
- The high frequency linear transducer is used for pneumothorax evaluation.
- The low frequency curved or phased array transducers are used for evaluations deep to the pleura.
- Ultrasound machine with B and M-modes.
2. Indications
- Evaluation of the lung for the presence of pneumothorax, pulmonary edema, consolidation, and pleural effusion.
- Evaluates the periphery of the lung rather than the central aspect of the lung.
- More sensitive in detecting pneumothorax, pulmonary edema, and consolidation compared to traditional chest x-ray.
- Can be performed quickly at the bedside while not exposing the patient to radiation.
- Scans can be repeated easily to evaluate the management of a disease process.
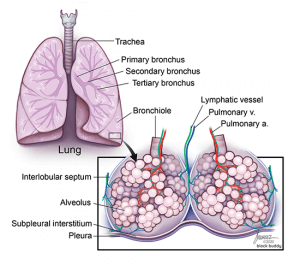
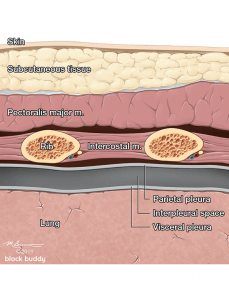
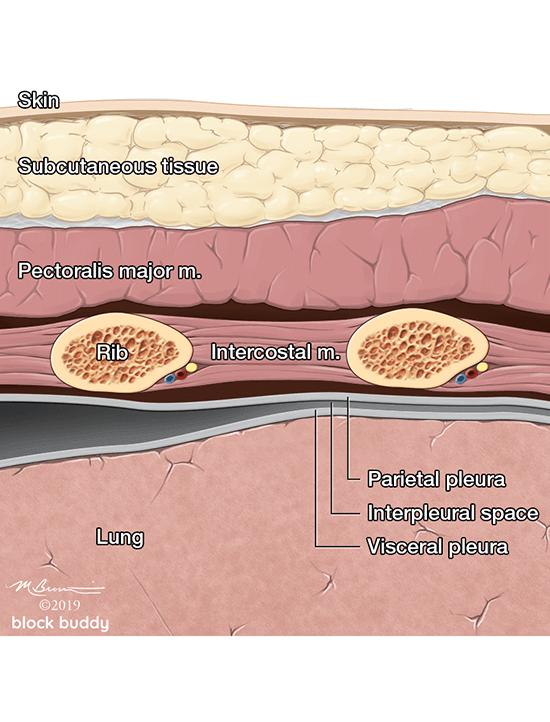
3. Anatomy
Secondary Pulmonary Lobule
- Located in the periphery of the lung.
- Comprised of 3 components: centrilobular, lobular parenchyma, and interlobular septa.
- Centrilobular: center portion of each lobule that is supplied by a respiratory bronchiole and pulmonary artery.
- These structures are surrounded by peribronchovascular interstitium.
- Lymph vessels run parallel to these structures.
- Lobular parenchyma: surrounds the centrilobular structures and contains the acini and intralobular interstitium.
- Acini: functional units of the lung that contains respiratory bronchioles, alveolar ducts, and alveoli.
- Intralobular interstitium: connective tissue around the alveoli that contains the pulmonary capillary bed.
- Interlobular septa: connective tissue that surrounds the lobular and centrilobular structures.
- Extend inward from subpleural interstitium and separate adjacent secondary pulmonary lobules.
- Contain pulmonary veins and lymph vessels.
- Interlobular septal thickening can occur from an increase in fluid within the septa (i.e. pulmonary edema).
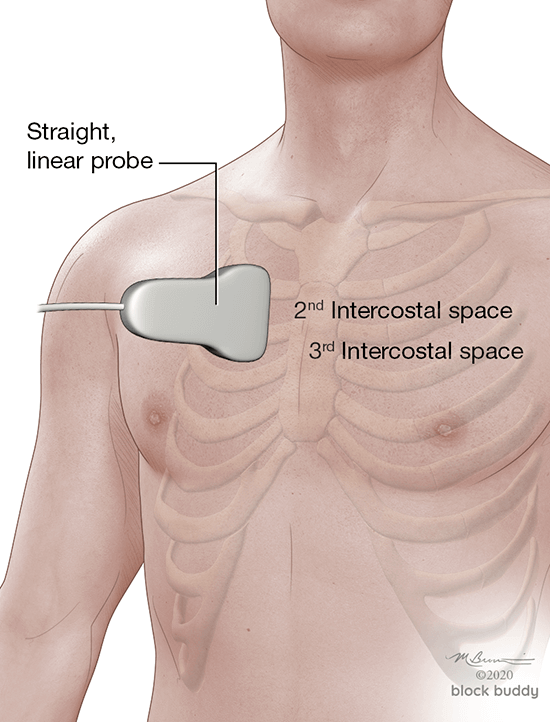
4. Positioning and Scanning
- Scanning can be performed with the patient in a supine, lateral or sitting position.
- In the critically ill patient, perform the scan with the patient supine and modified lateral position.
- When evaluating for a pneumothorax, perform the scan with the patient in the supine or semi-recumbent position.
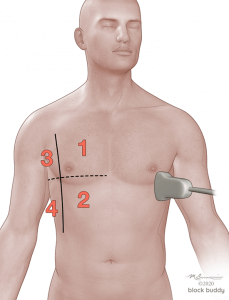
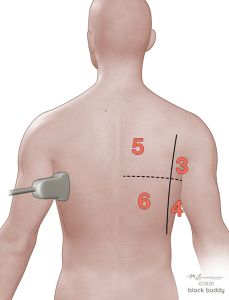
- Use the anterior axillary (AAL) and posterior axillary (PAL) lines to divide each hemithorax into an anterior, lateral, and posterior zone.
- Further divide each zone into a superior and inferior region by drawing an imaginary horizontal line.
- This will create 6 zones over each hemithorax.
- Initially place the transducer in the long axis over zone 1.
- Reposition the transducer over each subsequent zone to complete the evaluation.
5. Normal Lung Ultrasound
Anatomy
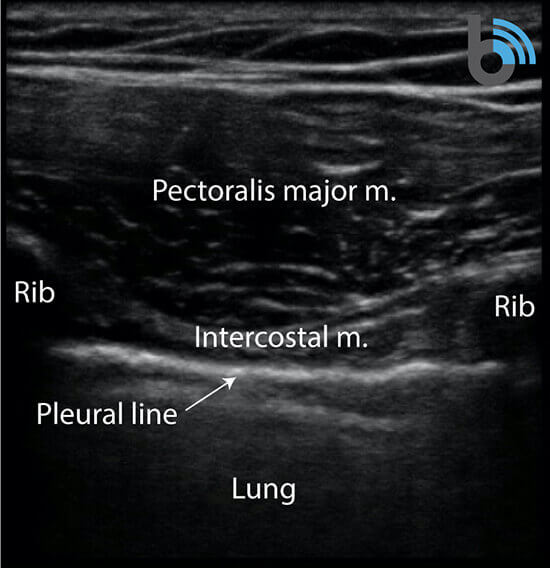
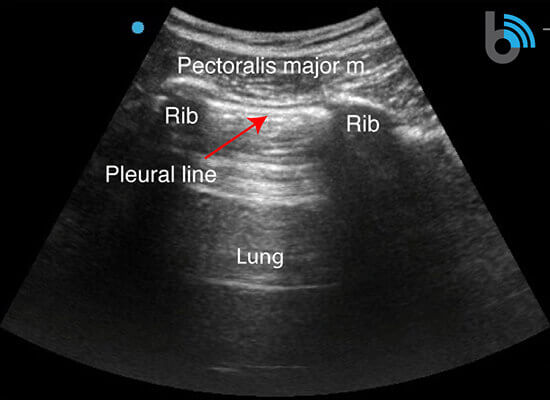
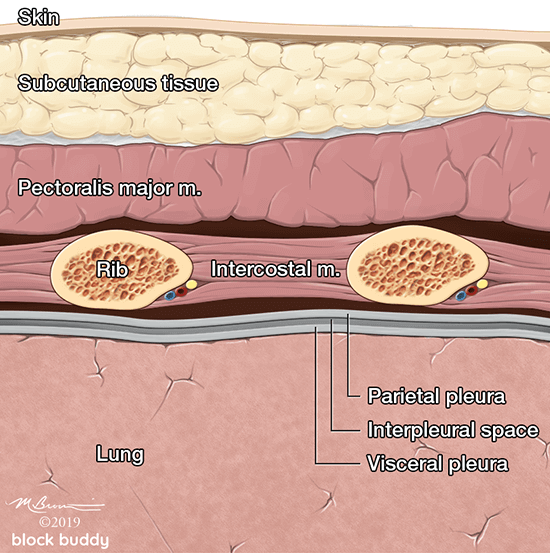
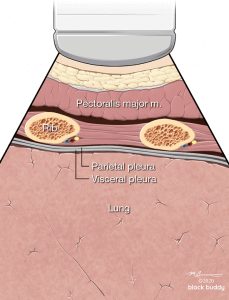
- The ribs will appear as convex hyperechoic lines with acoustic shadows.
- Located between the ribs are the hypoechoic intercostal muscles and the smooth, thin hyperechoic pleural line.
- These structures form the “bat sign”: the shadow of the ribs form the wings, while the area between the ribs represents the body.
Lung Sliding
- The pleural line is comprised of the parietal and visceral pleura, but will appear as a single, thin hyperechoic line.
- During normal respiration, horizontal lung sliding occurs as the parietal and visceral pleura slide in opposition.
- The pleural line between the ribs will appear to move or shimmer.
- Small vertical white lines (I-lines), also referred to as comet-tail artifacts, may extend from the pleura.
- These confirm the pleural layers are in contact.
- Helps exclude a pneumothorax.
- Easier to identify with high frequency transducers.
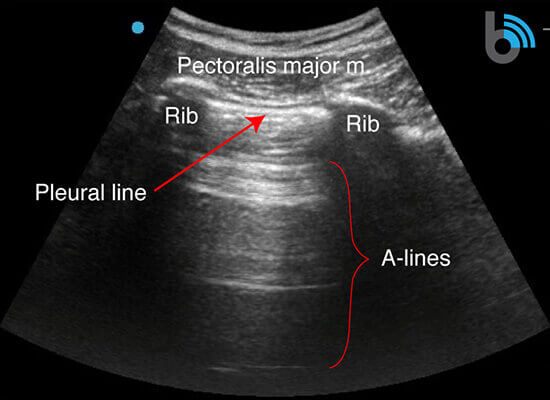
A-lines
- Hyperechoic, horizontal reverberation artifacts.
- Equally spaced
- Extend below the pleural line.
- Subpleural air (due to alveolar air or pneumothorax) creates a large difference in acoustic impedance at the interface between the chest wall and the air filled alveoli.
- Sound waves will reflect between the transducer and pleura several times, producing the repetitive lines.
- The appearance of A-lines indicates the presence of subpleural air, but does not differentiate between alveolar air (normal lung) and pleural air (pneumothorax).
- A-lines in the presence of lung sliding indicate alveolar air and not a pneumothorax.
- A-lines in the absence of lung sliding may indicate a pneumothorax, but requires additional findings (i.e. lung point).
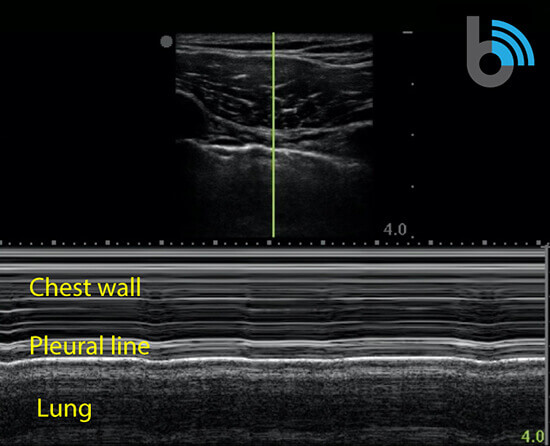
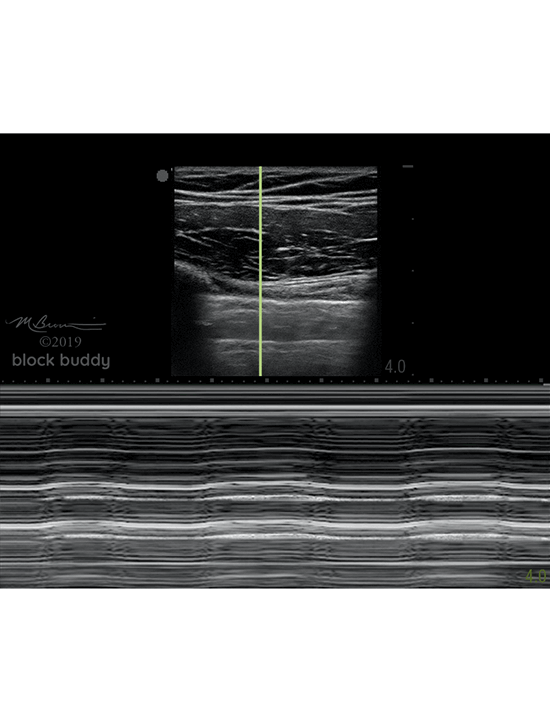
M-mode (motion mode)
- Measures the motion of a structure over a period of time.
- After obtaining a B-mode image, a single scan line (yellow line) is placed over the pleura between the ribs.
- A normal aerated lung will present as the “seashore sign” or “waves on the beach”.
- The “waves” will appear as horizontal, hyperechoic lines.
- Represents the lack of motion in the chest wall.
- The area below the pleural line will appear granular (“beach”)
- Results from the motion of the lung.
6. Abnormal Lung Ultrasound
B-Lines
- Hyperechoic, vertical reverberation artifacts.
- Extend from the pleural line to the bottom of the screen.
- Obliterate A-lines.
- Move synchronously with lung sliding.
- Results from interlobular septal thickening (fluid) surrounded by air filled alveoli.
- This produces a significant acoustic impedance gradient, resulting in the reverberation artifacts.
- Most commonly seen in pulmonary edema (cardiogenic and non-cardiogenic).
- Also seen with interstitial pneumonia, acute respiratory distress syndrome (ARDS), pulmonary fibrosis, pulmonary contusion, atelectasis and malignancy.
- 2 or less B-lines between 2 ribs in any zone are a normal finding.
- 3 or more B-lines between ribs in any zone are pathologic.
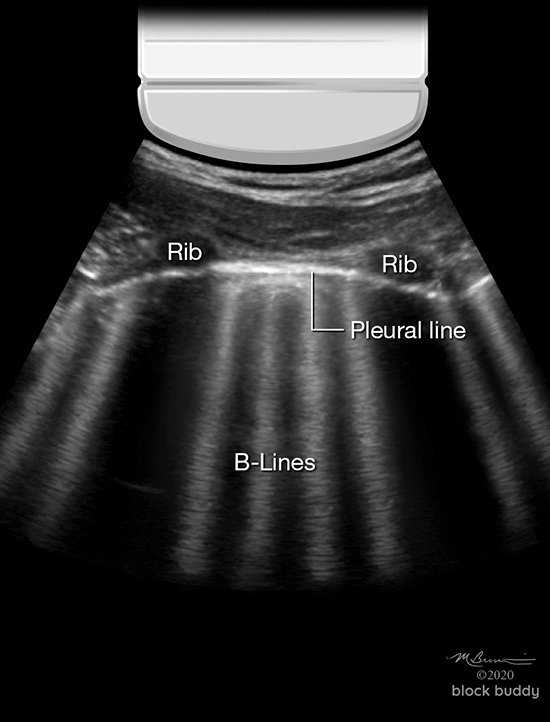
- The presence of B-lines can be used to identify interstitial syndrome, but cannot distinguish between pulmonary edema, ARDS, or fibrosis.
- B-lines with lung sliding are indicative of pulmonary edema.
- B-lines without lung sliding, may indicate ARDS or pneumonia.
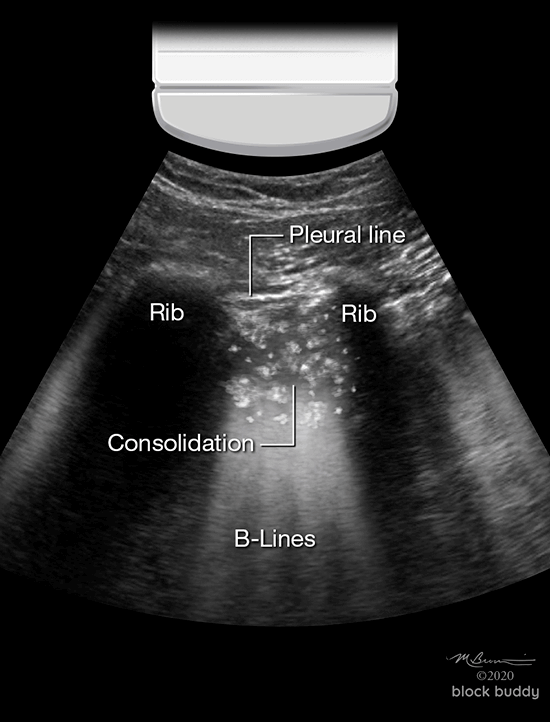
Consolidation
- Alveoli become filled with fluid: transudate, exudate, blood, or saline.
- Consolidation can be translobar or non-translobar.
- Translobar consolidation produces “tissue like sign” on ultrasound.
- Alveoli within entire lobe are filled with fluid.
- Causes hepatization: lung appears solid with a hypoechoic, homogenous echotexture similar to the liver.
- Lung dimensions are preserved.
- Non-translobar consolidation produces the “shred sign”.
- Most commonly seen consolidation.
- Involves a localized area or segment of the lobe.
- Within the lobe, there are areas of consolidation adjacent to normal aerated lung.
- The border between these areas area irregular and shredded.
- Commonly occur in the anterior chest.
- B-lines may extend from the shredded border to the bottom of the screen.
- Air bronchograms may be present in translobar or non-translobar consolidations.
- These hyperechoic specks or linear artifacts represent air within bronchi surrounded by fluid filled alveoli.
- These can be classified as static (motionless) or dynamic.
- Static air bronchograms indicate trapped air and do not move.
- Dynamic air bronchograms move with respiration and indicate the movement of air through fluid filled smaller airways.
- Consolidation most commonly occurs in patients with pneumonia.
- Patients with atelectasis may also present with a consolidation pattern on ultrasound.
Pleural Effusion
- Accumulation of fluid within the pleural space.
- Commonly occurs with congestive heart failure, infections (pneumonia), renal failure, malignancy, and cirrhosis.
- Ultrasound can detect as little as 5ml of pleural fluid, and is more sensitive, specific, and accurate than chest x-ray.
- Ultrasound can also be used to assess pleural fluid volume and perform a thoracentesis.
- Estimate effusion volume using the following formula:
- Effusion volume (ml) = distance between pleura and diaphragm (mm) x 20
- Scanning can be performed in the supine position with the head of bed elevated, allowing fluid to collect in the dependent area of the lower thorax above the diaphragm.
- The transducer is placed in the long axis of zone 4, at the level of the diaphragm, near the mid or posterior axillary line.
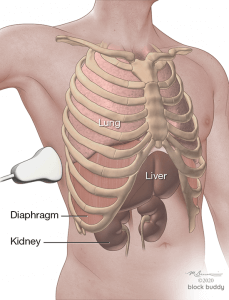
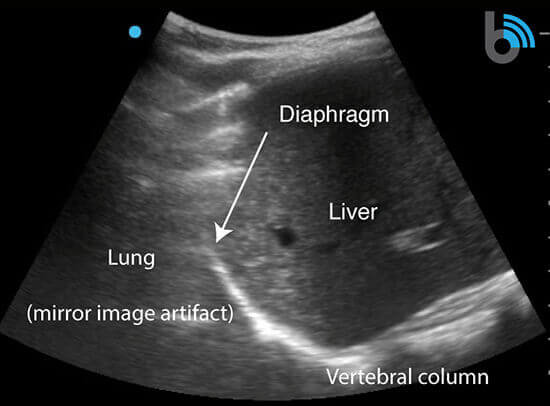
- A normal exam will reveal the liver/spleen, diaphragm, mirror image of the liver/spleen superior to the diaphragm, and the vertebral column lying posterior to the liver/spine (“spine sign”).
- The lung cannot be visualized due to the large acoustic impedance between the chest wall and air filled alveoli.
- The spine sign is only present posterior to the liver/spleen where ultrasound waves can pass through the structures to reach the vertebral column.
- The spine sign will not be present superior to the diaphragm, as the ultrasound waves are not transmitted through a normal aerated lung.
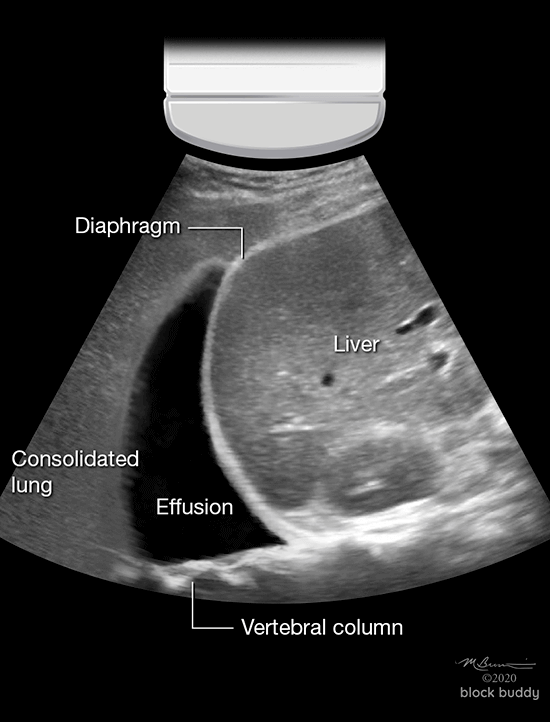
- In the presence of a pleural effusion, an anechoic collection of fluid will appear superior to the diaphragm.
- The lung will appear consolidated from pleural fluid compression.
- The spine sign can be viewed posterior to the pleural effusion (superior to the diaphragm) since fluid can transmit ultrasound waves to the vertebral column.
7. Pulmonary Pathology
COVID-19 and Lung Ultrasound
- Ultrasound findings (B-lines, consolidations, air bronchograms, and pleural abnormalities) are most commonly seen in the posterior lower fields of both lungs.
- Ultrasound is a quick and efficient tool that can be used at the bedside to detect pulmonary changes that may occur in COVID-19 patients.
- CXR and chest CT are non-specific and expose the patient to radiation.
- Ultrasound scans can be repeated easily to assess disease process.
- Due to the small size of the COVID-19 virus, the particles may be inhaled into the lungs and alveoli.
- Infection usually starts in the alveoli, close to the pleura, allowing for easy identification on ultrasound.
- Perform the scan using a low frequency curvilinear or phased array transducer.
- In addition, a high frequency linear transducer can be used to improve image resolution and enhance diagnostic accuracy of pleural abnormalities and parapneumonic effusions.
- The findings below are based on current knowledge and understanding of COVID-19.
- The findings may be variable, depending on the severity/stage of the disease process.
Mild COVID-19
- Normal or reduced pleural sliding
- Regular or mild pleural thickening
- Focal B-lines
- Mild pulmonary edema
Moderate to Severe COVID-19
- Pleural line is thickened, irregular and interrupted
- Multiple or confluent B-lines
- Subpleural consolidation may appear patchy or nodule
- Air bronchiologram or air bronchogram may be present within consolidation
- Color Doppler may show insufficient blood supply within consolidation
- Moderate to severe alveolar-interstitial pulmonary edema
- Large pleural effusion (rare)
Pneumonia
- Inflammatory infection process.
- Alveoli fill with inflammatory or purulent fluid.
- Lung sliding may be present or absent.
- Pleural line may be thick and irregular.
- B-lines are focal.
- Small subpleural consolidation with possible air bronchograms (static or dynamic) may be present.
- Dynamic air bronchograms are highly specific for pneumonia.
Atelectasis
- The alveoli are collapsed and there is a reduction in lung volume.
- Non-aerated lung will appear solid, similar to a consolidation.
- Use ultrasound and secondary findings to help differentiate between pneumonia and atelectasis:
- Air bronchograms with atelectasis are static and not dynamic.
- Large pleural effusions may indicate compressive atelectasis.
- Small pleural effusions are more likely to occur with pneumonia.
- Elevated white blood cell count and fever in the presence of consolidation may indicate pneumonia.
Pulmonary Edema
- Results from an increase of fluid in the lung interstitium.
- Lung sliding is present.
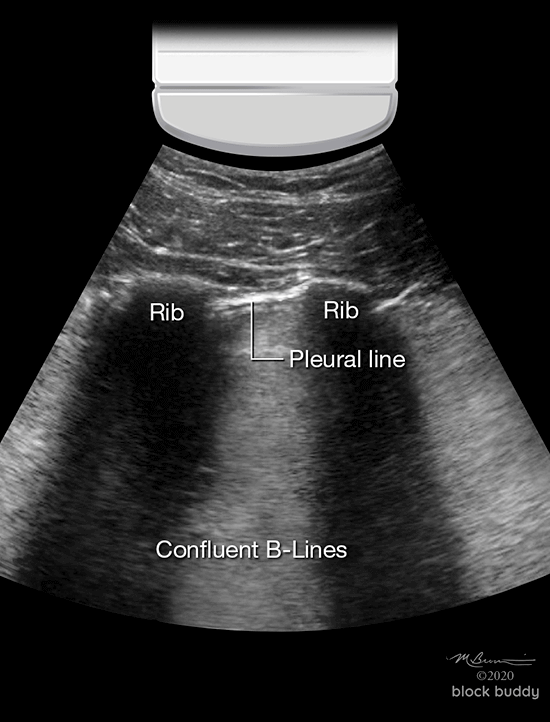
- 3 or more B-lines in at least 2 zones confirm the presence of pulmonary edema.
- Pulmonary edema is the most common cause of B-lines.
- As the severity of the edema progresses, multiple diffuse B-lines are produced.
- A hyperechoic confluent pattern (white out) between the ribs may occur as the B-lines fuse together.
Acute Respiratory Distress Syndrome (ARDS)
- Causes diffuse inflammation of lung tissue.
- Lung sliding is reduced or absent.
- B-lines are present with areas of sparing.
- Pleural line is thickened.
- Alveolar consolidation with air bronchograms may be present.
Pneumothorax
- The parietal and visceral pleura are separated by the collection of air within the pleural space.
- Lung sliding is absent, as air within the pleural space prevents the transmission of sound waves to the visceral pleura.
- Only the parietal pleura can be visualized.
- Scan the patient in a supine or semi-recumbent position.
- The transducer is positioned in the long axis over the most anterior aspect of the chest.
- Air within the pleura collects at the highest point within the chest.
- The ribs, interspace, and pleural line are identified.
- Absence of lung sliding does not confirm a pneumothorax.
- This can also occur in patients with right mainstem intubation, ARDS, COPD, asthma, pneumonia, pleural adhesions, effusions and subcutaneous emphysema.
- A-lines may appear, but cannot confirm the presence of a pneumothorax.
- These artifacts result from subpleural air (alveolar air or pneumothorax).
- Further evaluation using M-mode and lung point is required.
- B-lines are not present with a pneumothorax.
- M-mode can be used to measure the motion of the chest wall and lung.
- When a pneumothorax is present, the “bar code” sign will appear as horizontal, hyperechoic lines across the screen.
- Represents the lack of motion in the chest wall and lung.
- Lung point is the location at which normal sliding lung meets absent lung sliding (pneumothorax border).
- This can also be described as the point at which the parietal and visceral begin to separate in the presence of a pneumothorax
- Lung point appears in cases of incomplete pneumothorax.
- Identification of the lung point has been found to be 100% specific for confirming a pneumothorax.
- If lung sliding is not detected, the transducer is moved laterally until lung sliding is visualized.
- The lung point is then identified.
- The transducer can also be rotated transverse along the intercostal space to provide a better image of the pleural line and lung point.
8. Clinical Pearls
- When using a curved transducer, increase the depth to >10cm to determine if the comet-tail artifact is a B-line or I-line.
- B-lines will extend to the bottom of the screen.
- I-lines will only extend several centimeters below the pleural line.
- The absence of A-lines can occur if the transducer is not parallel to the pleura.
- Perform bilateral scanning to compare each hemithorax.
- Scanning multiple intercostal spaces increases sensitivity.
9. Video Gallery
Videos courtesy of John Shields, CRNA and Intelligent Ultrasound.
10. Illustrations
Our illustrations are completed by Joseph Kanasz, BFA, CMI of JK Illustration ([email protected]) and Matthew Brownstein, mb2media ( www.mb2media.com)
11. References
Huang Y, Wang S, Liu Y, Zhang Y, et al. A preliminary study on the ultrasonic manifestations of peripulmonary lesions of non-critical novel coronavirus pneumonia (COVID-19).
Peng QY, Wang XT, Zhang LN. Findings of lung ultrasonography of novel corona virus pneumonia during the 2019-2020 epidemic. Intensive Care Med, 2020; https://doi.org/10.1007/s00134-020-05996-6.
Lichtenstein DA, Meziere G. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: The BLUE Protocol. Chest. 2008;134(1):117-25.
Lichtenstein DA. BLUE-Protocol and FALLS-Protocol: Two Applications of Lung Ultrasound in the Critically Ill. Chest. 2015;147(6):1659-70. doi: 10.1378/chest.14-1313. PubMed PMID: 26033127.
Volpicelli G, Mussa A, Garofalo G, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med. 2006;24(6):689-96.
Volpicelli, Melniker LA, Cardinale L, et al. Lung ultrasound in diagnosing and monitoring pulmonary interstitial fluid. Radiol med. 2011.
Piette E, Daoust R, Denault A. Basic concepts in the use of thoracic and lung ultrasound. Curr Opin Anesthesiol. 2013;26(1);20-30.
Doerschug KC, Scmidt GA. Intensive Care Ultrasound: III. Lung and Pleural Ultrasound for the Intensivist. Ann Am Thorac Soc, 2013.
Baston C, Eoin West T. Lung ultrasound in acute respiratory distress syndrome and beyond. J Thorac Dis. 2016;8(12):E1763-E1766.
Deeb MA, Barbic S, Featherstone R, et al. Point-of-care ultrasonography for the diagnosis of acute cardiogenic pulmonary edema in patients presenting with acute dyspnea: a systematic review and meta-analysis. Acad Emerg Med. 2014; 21(8):844-52.
Durant A, Nagdev A. Ultrasound Detection of Lung Hepatization. West J Emerg Med. 2010;11(4):322-323.
Arabiat M, Foderaro AE, Levinson AT. Lung Ultrasound for Diagnosing Patients with Severe Dyspnea and Acute Hypoxic Respiratory Failure. R I Med J. 2019;102(10):34-38.
Dietrich CF, Mathis G, Blaivas M, et al. Lung B-line artefacts and their use. J Thorac Dis. 2016;8(6):1356-1365.
Lichtenstein D, Ambroise Pare H. Ultrasound diagnosis of atelectasis. International Journal of Intensive Care. 2005;12(2):88-93.
Gillman LM, Panebianco N, Alkadi A, et al. The dynamic sonographic air bronchogram: a simple and immediate bedside diagnosis of alveolar consolidation in severe respiratory failure. J Trauma. 2011;70(3):760.
Lichtenstein D, Meziere G, Seitz J. The dynamic air bronchogram. A lung ultrasound sign of alveolar consolidation ruling out atelectasis. Chest. 2009;135(6):1421-1425.
Miller A. Practical approach to lung ultrasound. BJA Education. 2016;16(2):39-45.
Sikora K, Perera P, Mailhot T, Mandavia D. Ultrasound for the Detection of Pleural Effusions and Guidance of the Thoracentesis Procedure. ISRN Emergency Medicine. 2012;doi:10.5402/2012/676524.
Lichtenstein D. Lung ultrasound in the critically ill. Ann Intensive Care.2014;4:1.
Perrone T, Maggi A, Sgarlata C, et al. Lung ultrasound in internal medicine: A bedside help to increase accuracy in the diagnosis of dyspnea. Eur J Intern Med. 2017;46:61-65.
Zanobetti M, Scorpiniti M, Gigli C, et al. Point-of-Care Ultra- sonography for Evaluation of Acute Dyspnea in the ED. Chest. 2017;151(6):1295-1301.
Lichtenstein DA, Meziere GA, Lagoueyte JF, Biderman P, Gold- stein I, Gepner A. A-lines and B-lines: lung ultrasound as a bed- side tool for predicting pulmonary artery occlusion pressure in the critically ill. Chest. 2009;136(4):1014-1020.
Lichtenstein D, Meziere G, Biderman P, Gepner A. The com- et-tail artifact: an ultrasound sign ruling out pneumothorax. Intensive Care Med. 1999;25(4):383-388.
Corradi F, Brusasco C, Pelosi P. Chest ultrasound in acute re- spiratory distress syndrome. Curr Opin Crit Care. 2014;20(1): 98-103.
